Oracle IAS, the best coaching institute for UPSC/IAS/PCS preparation in Dehradun brings to you UKPCS Science Chemistry (paper 6) –
Steel is an alloy of iron and other elements, primarily carbon that is widely used in construction and other applications because of its high tensile strength and low cost. Steel’s base metal is iron. The carbon content of steel is between 0.002% and 2.1% by weight for plain iron–carbon alloys. These values vary depending on alloying elements such as manganese, chromium, nickel, iron, tungsten, carbon and so on. Today, it is one of the most common materials in the world, with more than 1.3 billion tons produced annually. It is a major component in buildings, infrastructure, tools, ships, automobiles, machines, appliances, and weapons.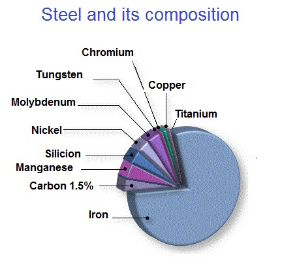
Steel making is the process for producing steel from iron ore and scrap. In steel making, impurities such as nitrogen, silicon, phosphorus, sulfur and excess carbon are removed from the raw iron, and alloying elements such as manganese, nickel, chromium and vanadium are added to produce different grades of steel.
Modern steel making processes can be broken into two categories: primary and secondary steel making. Primary steel making involves converting liquid iron from a blast furnace and steel scrap into steel via basic oxygen steel making or melting scrap steel and/or direct reduced iron (DRI) in an electric arc furnace. Secondary steel making involves refining of the crude steel before casting and the various operations are normally carried out in ladles.
PRC, EU, Japan, India and US are the largest producer in the world. India has replaced Japan as world’s second largest producing country, while China is the largest producer of crude steel accounting for more than 51 per cent of production.
Printed notes topic wise – Contact us
Cost- Rs.5000/- (including shipping)
(~2500+ pages)
UKPCS Mains Study Material subject wise
The notes are strictly as per UKPCS syllabus (topic wise):
Individual Polity Cost: Rs. 1500/- (including shipping)
Individual S&T Cost: Rs. 1500/- (including shipping)
Individual Geography Cost: Rs. 1500/- (including shipping)
Individual Economics Cost: Rs. 1000/- (including shipping)
Individual Ethics Cost: Rs. 1000/- (including shipping)
Individual History Cost: Rs. 1500/- (including shipping)
Contact us for:-
-
-
-
-
-
-
- Upper Mains UKPCS Classroom program
- UKPCS Upper Test Series
- UKPCS Lower Test Series
- UKPCS Lower Classes
- UKPCS Past year papers
- Phone Number:–9997453844
- Telegram channel : click here
-
-
-
-
-
- UKPSC अपर प्रीलिम्स 2026 टेस्ट सीरीज़ || Oracle IAS - April 10, 2026
- Master Uttarakhand Current Affairs for UKPCS: CM Samvad & Monthly PDF Download - February 20, 2026
- UKPSC प्रारंभिक परीक्षा स्टडी मटेरियल 2026 (Upper & Lower) | Oracle IAS - January 3, 2026
